Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Nakashima, Nobuaki*; Yatsuhashi, Tomoyuki*; Sakota, Kenji*; Iwakura, Izumi*; Hashimoto, Sena*; Yokoyama, Keiichi; Matsuda, Shohei
Chemical Physics Letters, 752, p.137570_1 - 137570_5, 2020/08
Times Cited Count:1 Percentile:5(Chemistry, Physical)Photo-redox reactions between Eu and Eu
and Eu ions are induced by laser irradiation in alcoholic solution. Efficiency, wavelength dependence, and laser-power dependence are investigated with three different lasers. Nano second laser pulses at a wavelength of 308 nm is found to cause one-photon redox reactions with a quantum yield around 0.5. Nano second laser pulses at a wavelength of 394 nm induces two-photon reduction of Eu
ions are induced by laser irradiation in alcoholic solution. Efficiency, wavelength dependence, and laser-power dependence are investigated with three different lasers. Nano second laser pulses at a wavelength of 308 nm is found to cause one-photon redox reactions with a quantum yield around 0.5. Nano second laser pulses at a wavelength of 394 nm induces two-photon reduction of Eu to form Eu
to form Eu . When the pulse energy is 5 mJ, the quantum yield is measured to be 0.015. Although the quantum yield is one order of magnitude lower than that of the one photon reduction, reduction phenomena can be easily observed under the moderate laser field strength. Because of the two-photon nature, there should be a room to improve the efficiency by increasing the laser field strength.
. When the pulse energy is 5 mJ, the quantum yield is measured to be 0.015. Although the quantum yield is one order of magnitude lower than that of the one photon reduction, reduction phenomena can be easily observed under the moderate laser field strength. Because of the two-photon nature, there should be a room to improve the efficiency by increasing the laser field strength.
Ozaki, Takuo; Kimura, Takaumi; Onuki, Toshihiko; Francis, A. J.
Radiochimica Acta, 94(9-11), p.715 - 721, 2006/11
Times Cited Count:10 Percentile:57.07(Chemistry, Inorganic & Nuclear)no abstracts in English
Ozaki, Takuo; Kimura, Takaumi; Onuki, Toshihiko; Yoshida, Zenko; Francis, A. J.
Environmental Toxicology and Chemistry, 22(11), p.2800 - 2805, 2003/11
Times Cited Count:21 Percentile:45.2(Environmental Sciences)no abstracts in English
Ozaki, Takuo; Kimura, Takaumi; Yoshida, Zenko; Francis, A. J.*
Chemistry Letters, 32(7), p.560 - 561, 2003/07
Times Cited Count:5 Percentile:26.49(Chemistry, Multidisciplinary)no abstracts in English
Suzuki, Hideya*; Naganawa, Hirochika; Tachimori, Shoichi
Solvent Extraction and Ion Exchange, 21(4), p.527 - 546, 2003/03
Times Cited Count:12 Percentile:45.03(Chemistry, Multidisciplinary)no abstracts in English
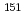 Eu M
Eu M ssbauer spectroscopic study on the Eu
ssbauer spectroscopic study on the Eu M
M O
O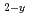 (0
(0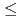 y
y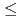 1.0)(M=Th, U)
1.0)(M=Th, U)Masaki, Nobuyuki; Otobe, Haruyoshi; Nakamura, Akio; Harada, Daijitsu*; Ito, Kentaro*; Sasaki, Yoshinobu*; Hinatsu, Yukio*
Journal of Nuclear Science and Technology, 39(Suppl.3), p.217 - 220, 2002/11
no abstracts in English
Kimura, Takaumi; Nagaishi, Ryuji; Arisaka, Makoto*; Ozaki, Takuo; Yoshida, Zenko
Radiochimica Acta, 90(9-11), p.715 - 719, 2002/11
Times Cited Count:15 Percentile:67.86(Chemistry, Inorganic & Nuclear)no abstracts in English
Nagaishi, Ryuji; Kimura, Takaumi; Yoshida, Yoichi*; Kozawa, Takahiro*; Tagawa, Seiichi*
Journal of Physical Chemistry A, 106(39), p.9036 - 9041, 2002/10
Times Cited Count:3 Percentile:8.65(Chemistry, Physical)no abstracts in English
Kimura, Takaumi; Nagaishi, Ryuji; Kato, Yoshiharu; Yoshida, Zenko
Journal of Alloys and Compounds, 323-324(1-4), p.164 - 168, 2001/07
Times Cited Count:50 Percentile:87.64(Chemistry, Physical)no abstracts in English
; ;
JNC TN8400 2001-022, 60 Pages, 2001/03
A numerical simulation code for the TRUEX (Transuranium Extraction) process was developed. Concentration profiles of americium and europium were calculated for some experiments of the counter current extraction system those were carried out in CPF (Chemical Processing Facility) by using the code. Calculation profiles were in agreement with the experimental results. Operational conditions were also examinted for the americium recovery experiment by the TRUEX process carried out in the Plutonium Fuel Center. It was shown that lowering the concentration of nitric acid in the scrub solution and decreasing the flow rate of solvent and strip solution was effective for improving the performance of the stripping step and reducing the volume of the waste solution. In order to find the optimum conditions for various experiments, this simulation code was modified to calculate the concentration profiles of other metal elements such as zirconium and iron and the effect of oxalic acid on the extraction behavior of the metal elements. The calculated concentration profiles of americium and europium were varied by this modification. In the experiment at CPF, the calculations were carried out to obtain recovery ratio of americium in the product stream with the amount of oxalic acid added to the process. This calculation result showed that it was possible to improve the performance of decontamination of fission products by increasing oxalic acid concentration added to the process. The calculation was also carried out for finding the optimum conditions of oxalic acid concentration added to the europium recovery process.
Takahashi, Yoshio*; Tada, Akisa*; Kimura, Takaumi; Shimizu, Hiroshi*
Chemistry Letters, (6), p.700 - 701, 2000/06
no abstracts in English
Osaka, Masahiko; Koyama, Shinichi; Mitsugashira, Toshiaki; Morozumi, Katsufumi; Namekawa, Takashi
JNC TN9400 2000-058, 49 Pages, 2000/04
The analytical technique for Cm contained in a MOX FUEL was developed and analysis of Cm contained in irradiated fuel of experimental fast reactor "JOYO" was carried out, to contribute to evaluation of transmutation characteristics of MA nuclide in the fast reactor. The procedure of ion-exchange separation of Cm with nitric acid-methanol mixed media essential for the isotopic analysis in irradiated MOX fuel was adopted considering for being rapid and easy. The fundamental test to grasp separation characteristics of this procedure, such as Cm elution position and separation capacity between Cm and Am or Eu, was carried out. ln applying this procedure to the analysis of Cm contained in actual specimen, separation condition was evaluated and optimized, and the procedure consist of impurity removal and Am removal process was devised. This procedure resulted in high recovery rate of Cm and high removal rate of Am and impurity which becomes a problem in sample handling and mass-spectrometry such as Eu and Cs. The Cm separation test from irradiated MOX fuel was carried out using this technique, and Cm isotopic ratio analysis was enabled. The analytical technique for Cm contained in irradiated MOX fuel was established using the procedure of ion-exchange separation with nitric acid-methanol mixed media. The analysis of Cm contained in irradiated MOX fuel of experimental fast reactor "Joyo" was carried out. As a result, it was revealed from measured data that Cm content rate was 1.4 4.0
4.0 lO
lO atom%, small amount of
atom%, small amount of 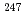 Cm was generated and Cm isotopic ratio was constant above burn-up 60GWd/t.
Cm was generated and Cm isotopic ratio was constant above burn-up 60GWd/t.
Naganawa, Hirochika; Suzuki, Hideya; Tachimori, Shoichi; Nasu, Akinobu*; Sekine, Tatsuya*
Bulletin of the Chemical Society of Japan, 73(3), p.623 - 630, 2000/03
Times Cited Count:6 Percentile:35.36(Chemistry, Multidisciplinary)no abstracts in English
Takahashi, Yoshio*; Kimura, Takaumi; Kato, Yoshiharu; Minai, Yoshitaka*
Environmental Science & Technology, 33(22), p.4016 - 4021, 1999/00
Times Cited Count:46 Percentile:74.45(Engineering, Environmental)no abstracts in English
Takahashi, Yoshio*; Kimura, Takaumi; Kato, Yoshiharu; *; *; *
Journal of Radioanalytical and Nuclear Chemistry, 239(2), p.335 - 340, 1999/00
Times Cited Count:15 Percentile:72.26(Chemistry, Analytical)no abstracts in English
Kimura, Takaumi; Kato, Yoshiharu
Journal of Alloys and Compounds, 275-277, p.806 - 810, 1998/00
Times Cited Count:110 Percentile:96.67(Chemistry, Physical)no abstracts in English
Kimura, Takaumi; Kato, Yoshiharu
Journal of Alloys and Compounds, 278, p.92 - 97, 1998/00
Times Cited Count:69 Percentile:92.91(Chemistry, Physical)no abstracts in English
Kimura, Takaumi; Kato, Yoshiharu; Takeishi, Hideyo; G.R.Choppin*
Journal of Alloys and Compounds, 271-273, p.719 - 722, 1998/00
Times Cited Count:48 Percentile:88.91(Chemistry, Physical)no abstracts in English
Takahashi, Yoshio*; *; Kimura, Takaumi; *
Journal of Radioanalytical and Nuclear Chemistry, 234(1-2), p.277 - 282, 1998/00
Times Cited Count:37 Percentile:91.63(Chemistry, Analytical)no abstracts in English